Additive Manufacturing has lead to advances in the medical field with applications in implantable medical devices. This technology, more commonly known as 3D printing, uses CAD drawings to create structures with intricate detail specific to the receiving patient. Previous to the introduction of 3D printing in the medical field, medical devices have been primarily created using compression molds, which are mass produced and non specific. There have been numerous stories in the media about implantable, 3D printed medical devices, but the short term and long term effects of these devices on human cells have not been yet explored. These products must be studied to ensure they are not only biocompatible, but will maintain structural integrity and strength as compared to their traditional molded counterparts.
In this study, traditionally molded samples were compared to 3D printed samples through mechanical, chemical, and thermal evaluation techniques.

(R) Traditionally molded ASTM dogbones

(R) High Resolution
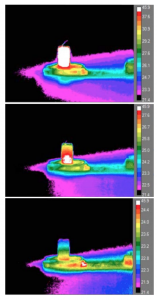
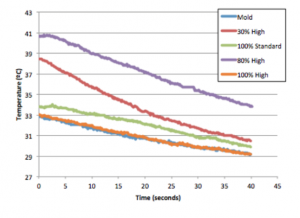
Samples created to test thermal dissapation during printing process.
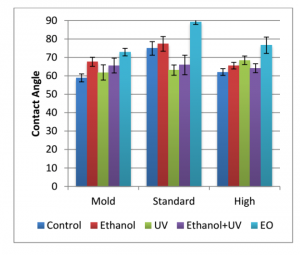
The effects of medical grade sterilization (ethanol, ultraviolet light, and ethylene oxide) on said properties were also explored. Polylactic acid (PLA) was used due to it’s current popularity in current medical devices, and because it has been one of the most explored materials in Fused Deposition Modeling (FDM) printing technology.


a- unsterilized
b- ethanol
c- UV
d- EtOH/UV
3D Printed samples had a higher average Young’s moduli of elasticity, indicating they are more brittle than their molded counterparts. Exposure to cell growth media decreased moduli readings and increased flexibility of the sample.
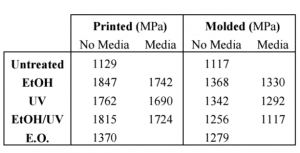
The infill of the sample (controlled by the user) had a direct correlation with heat dissipation; the lower the infill, shorter amount of time was needed for heat to dissipate throughout the sample. Sterilization altered the contact angle, and degraded the top layer of PLA, but did not significantly alter the moduli data. Ethylene oxide treated samples were shown to have the highest change in contact angle readings, indicating a more hydrophobic surface in both molded and printed samples.